It provides insights into the stability, reactivity, and bonding tendencies of different elements. For example, in the case of potassium, the single valence electron in the 4s orbital is easily lost, leading to the formation of a potassium ion (K+).īy understanding the electron configuration of an atom, scientists can predict and explain various chemical behaviors. These valence electrons are involved in bonding and determine the reactivity of an atom. The arrangement of electrons in the outermost energy level, known as the valence electrons, is particularly significant. It directly influences how atoms interact with one another to form compounds and participate in chemical reactions. The electron configuration of an atom plays a crucial role in determining its chemical properties. Importance of Electron Configuration in Chemical Properties This means that the first two energy levels (1s and 2s) are already filled with electrons from the argon atom, and the remaining electron occupies the 4s orbital of the third energy level. The electron configuration of potassium is 4s1, where represents the noble gas configuration of argon (1s2 2s2 2p6 3s2 3p6). To illustrate this, let’s take the example of the electron configuration of the potassium atom. Hund’s rule further states that electrons will occupy separate orbitals within the same subshell before pairing up. 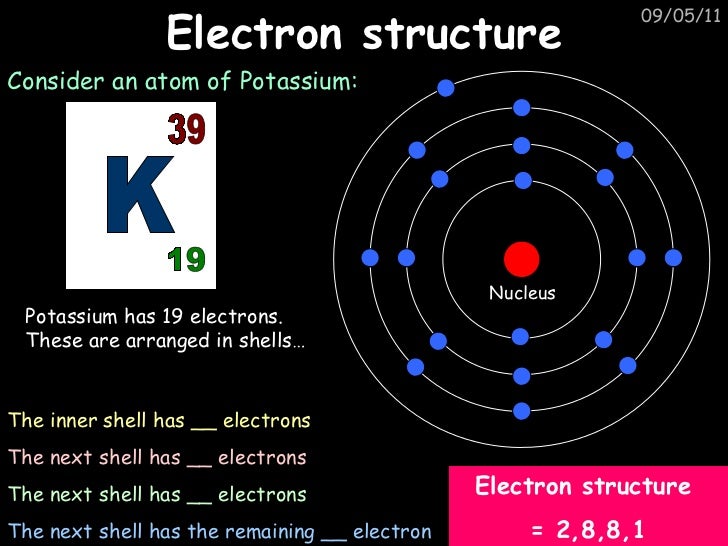
The Aufbau principle states that electrons fill the lowest energy orbitals first, while the Pauli exclusion principle states that each orbital can hold a maximum of two electrons with opposite spins. It is based on the concept of subshell configuration, which describes the filling of atomic orbitals with electrons. The electron configuration notation uses the periodic table as a guide to represent the electron arrangement of an atom. 
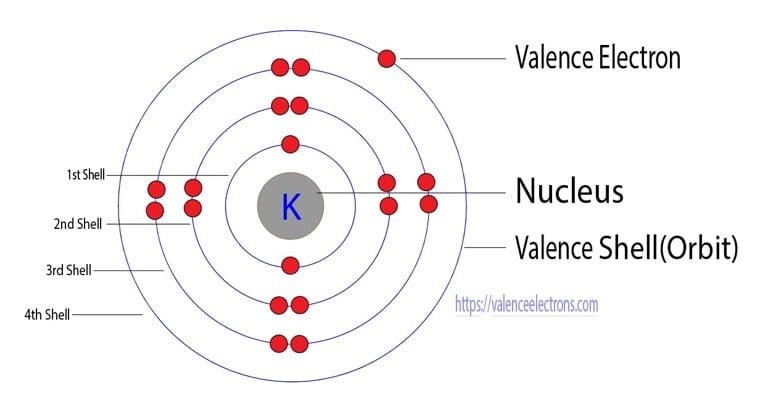
The electron distribution in these energy levels follows specific rules and patterns. In the atomic structure of potassium, for example, with an atomic number of 19, the electron configuration diagram shows that it has three energy levels: the first level can hold up to 2 electrons, the second level can hold up to 8 electrons, and the third level can hold up to 8 electrons as well. The electron configuration of an atom is determined by the principles of quantum mechanics and the properties of atomic orbitals. It provides a way to describe the distribution of electrons in different energy levels and orbitals. Key Takeaways: Atomic Number Symbol Electron Configuration 19 K 1s^2 2s^2 2p^6 3s^2 3p^6 4s^1 Understanding Electron Configuration Definition and Explanation of Electron ConfigurationĮlectron configuration refers to the arrangement of electrons within an atom. Understanding the electron configuration of potassium helps us comprehend its reactivity and its role in various chemical reactions. This means that potassium has a total of 19 electrons, with two in the 1s orbital, two in the 2s orbital, six in the 2p orbital, two in the 3s orbital, six in the 3p orbital, and one in the 4s orbital. The electron configuration of potassium is 1s^2 2s^2 2p^6 3s^2 3p^6 4s^1. One important aspect of potassium is its electron configuration, which determines its chemical behavior. It belongs to the alkali metal group and is highly reactive, readily forming compounds with various elements. Potassium is a chemical element with the symbol K and atomic number 19.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
